4:34 (Triple only) know that carboxylic acids contain the functional group -COOH
Carboxylic acids contain the functional group -COOH
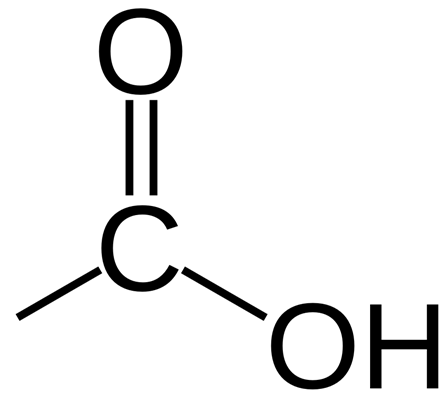
An example of a carboxylic acid is butanoic acid:
Carboxylic acids contain the functional group -COOH

An example of a carboxylic acid is butanoic acid:
The four simplest carboxylic acids are methanoic acid, ethanoic acid, propanoic acid and butanoic acid.
Methanoic acid
Displayed formula:
Molecular formula: CH₂O₂
Structural formula: HCOOH
Ethanoic acid
Displayed formula: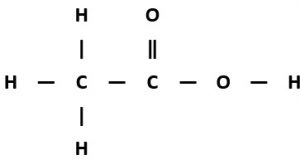
Molecular formula: C₂H₄O₂
Structural formula: CH₃COOH
Propanoic acid
Displayed formula:
Molecular formula: C₃H₆O₂
Structural formula: CH₃CH₂COOH
Butanoic acid
Displayed formula:
Molecular formula: C₄H₈O₂
Structural formula: CH₃CH₂CH₂COOH
Dilute carboxylic acids react with metals in the same way as other dilute acids (e.g. hydrochloric acid) only more slowly.
For example, dilute ethanoic acid reacts with magnesium with a lot of fizzing to produce a salt and hydrogen, leaving a colourless solution of magnesium ethanoate:
magnesium + ethanoic acid → magnesium ethanoate + hydrogen
Mg (s) + 2CH₃COOH (aq) → (CH₃COO)₂Mg (aq) + H₂ (g)
Dilute carboxylic acids react with metal carbonates as they do with other acids, to give a salt, carbon dioxide and water.
For example, dilute ethanoic acid reacts with sodium carbonate with a lot of fizzing to produce a salt, carbon dioxide and water, leaving a colourless solution of sodium ethanoate:
sodium carbonate + ethanoic acid → sodium ethanoate + carbon dioxide + water
Na₂CO₃ (s) + 2CH₃COOH (aq) → 2CH₃COONa (aq) + CO₂ (g) + H₂O (l)
As can be seen in the examples above the charge on the ethanoate ion is -1.
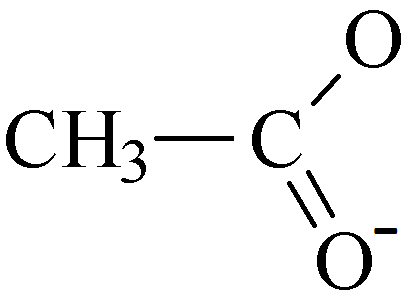
Vinegar is an aqueous solution containing ethanoic acid (CH₃COOH).