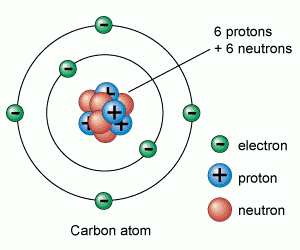
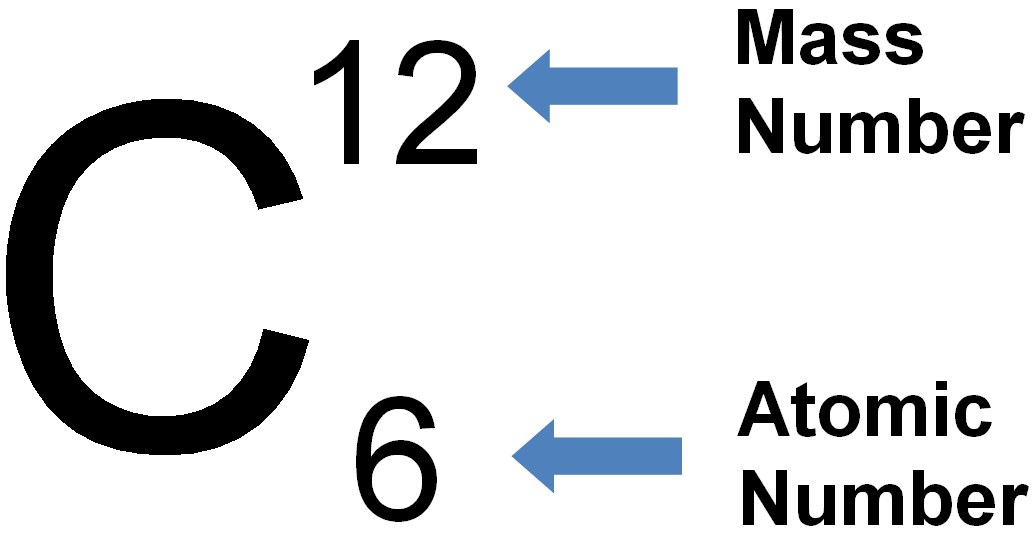
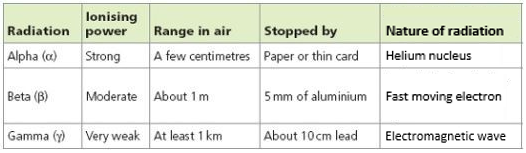
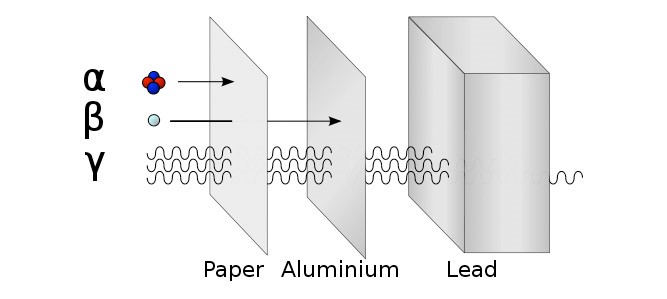
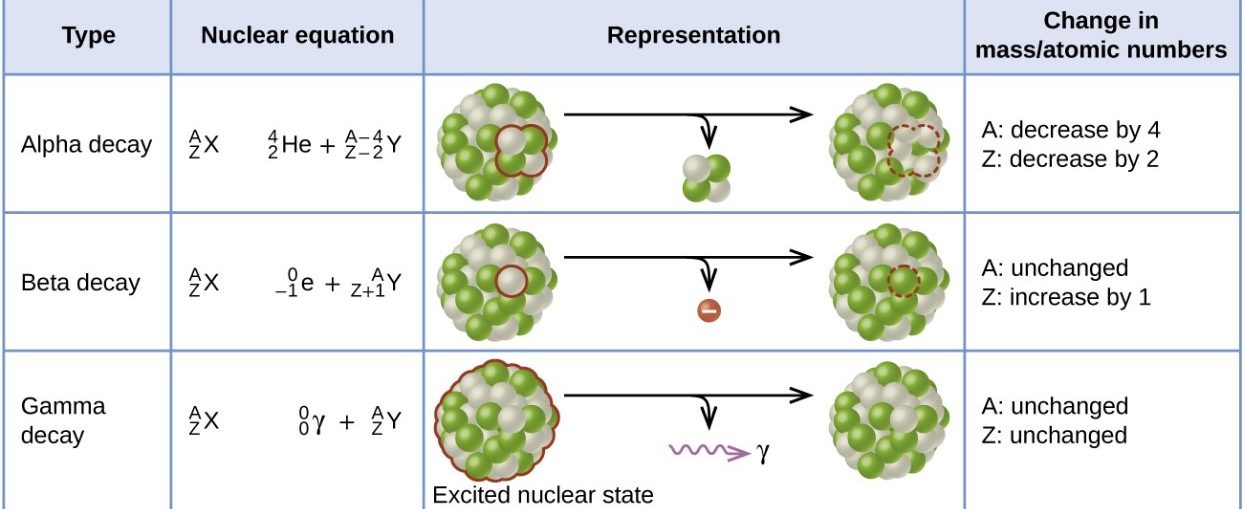
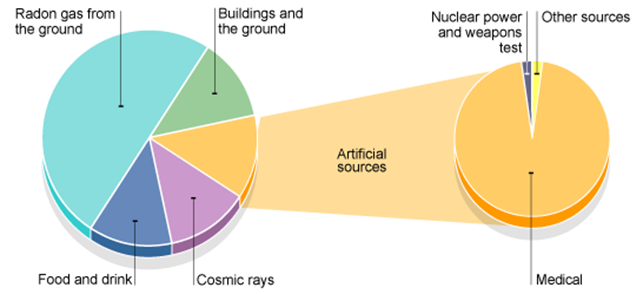
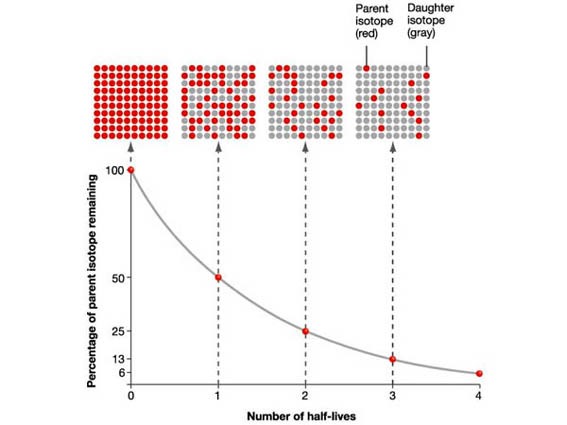
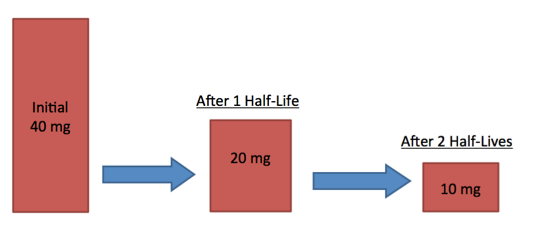
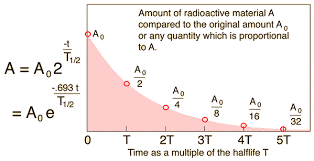
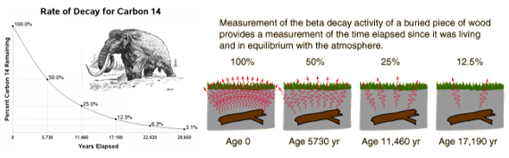
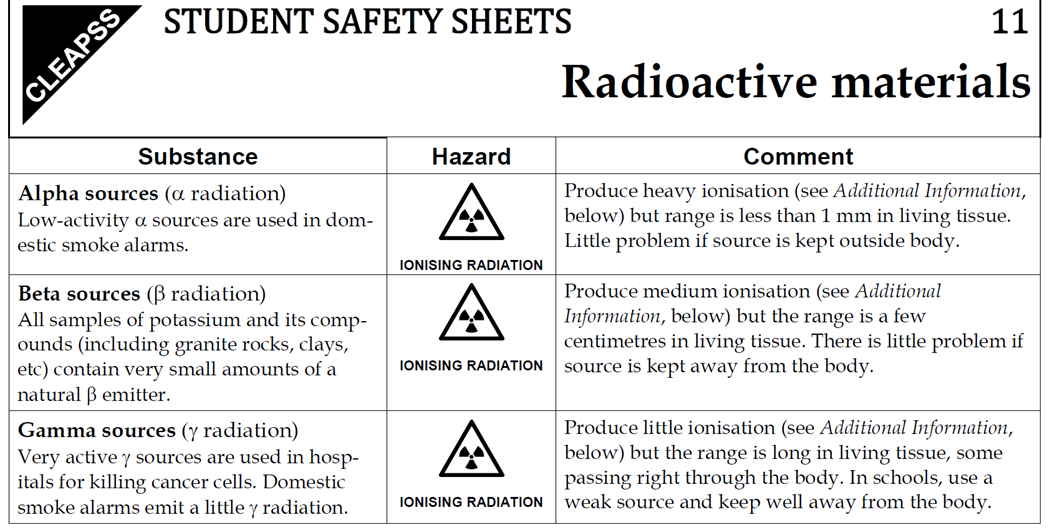
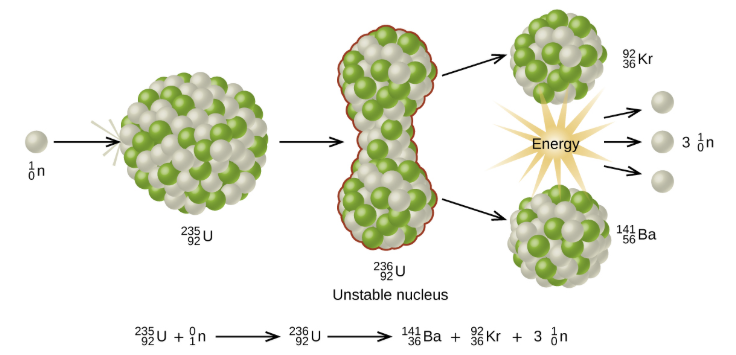
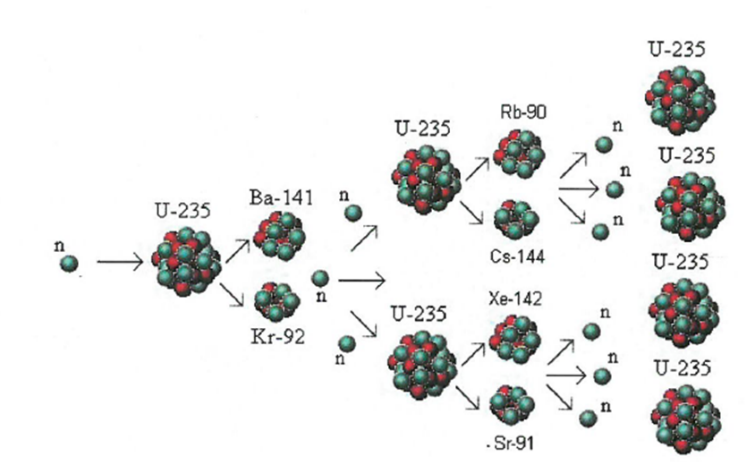
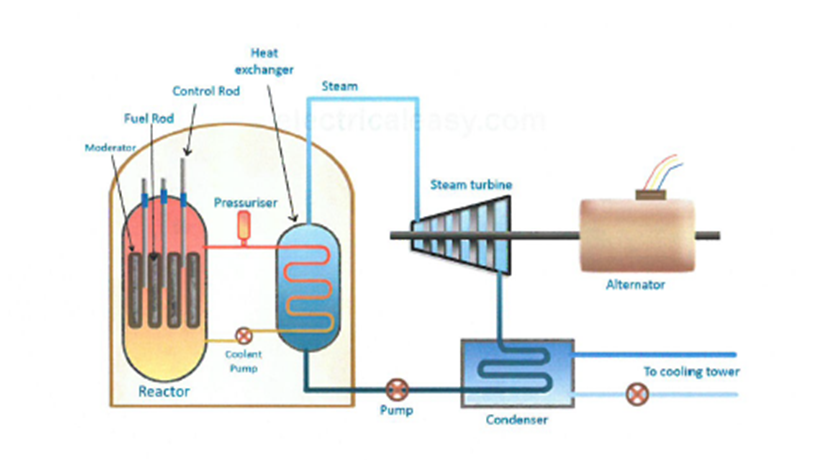
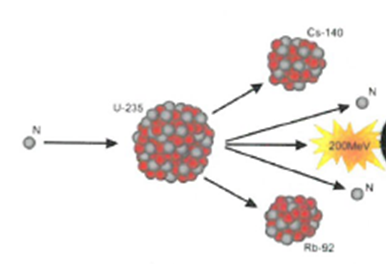
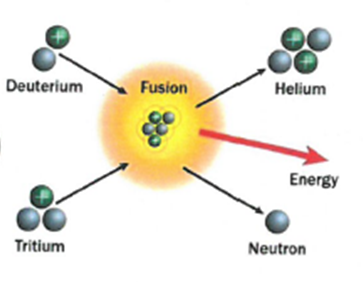
7.01 use the following units: becquerel (Bq), centimetre (cm), hour (h), minute (min) and second (s)
the units for:
frequency of decay : becquerel (Bq), 1 (Bq) for 1 decay / sec
distance : centimetres (cm), normally however is (m)
time : hour (h), minute (min) but normally (s)