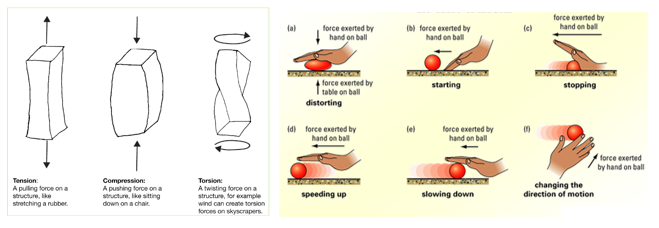
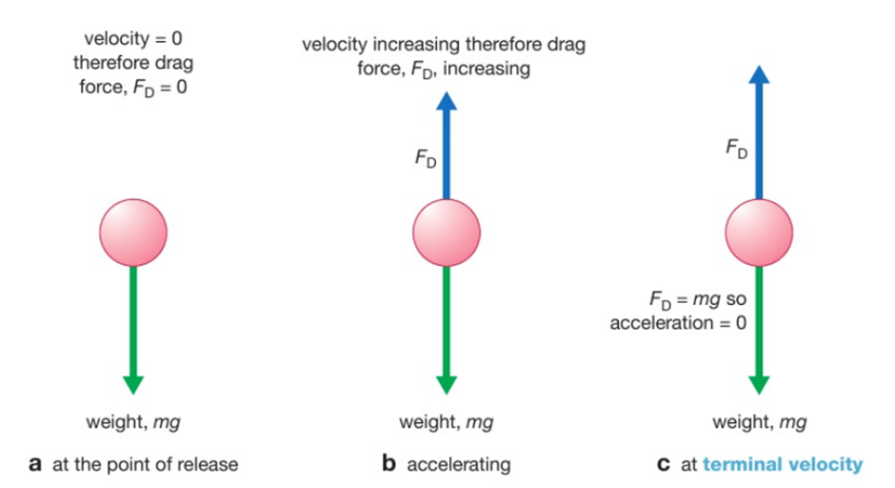
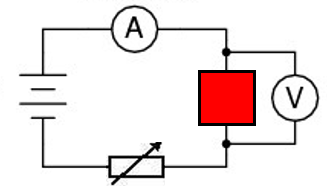
A flame test is used to show the presence of certain metal ions (cations) in a compound.
- A platinum or nichrome wire is dipped into concentrated hydrochloric acid to remove any impurities.
- The wire is dipped into the salt being tested so some salt sticks to the end.
- The wire and salt are held in a non-luminous (roaring) bunsen burner flame.
- The colour is observed.
Properties of the platinum or nichrome wire is:
- Inert
- High melting point