4.11 know and use the relationship between work done, force and distance moved in the direction of the force: W = F × d
Work Done (J) = Force (N) x distance moved (m)
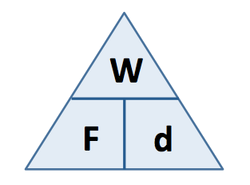
Work Done (J) = Force (N) x distance moved (m)

|
Work done = energy transferred |
Gravitational potential energy (J) = Mass (kg) x gravitational field strength (N/kg) x height (m)
Kinetic energy (J) = 0.5 x mass (kg) x velocity (m/s) 2
Because energy is conserved the decrease in GPE = increase in KE, for a falling object if no energy is lost to the surroundings
power is the rate of transfer of energy, or the rate of work done. so p = E/t